I know, it’s difficult to understand this statement. 
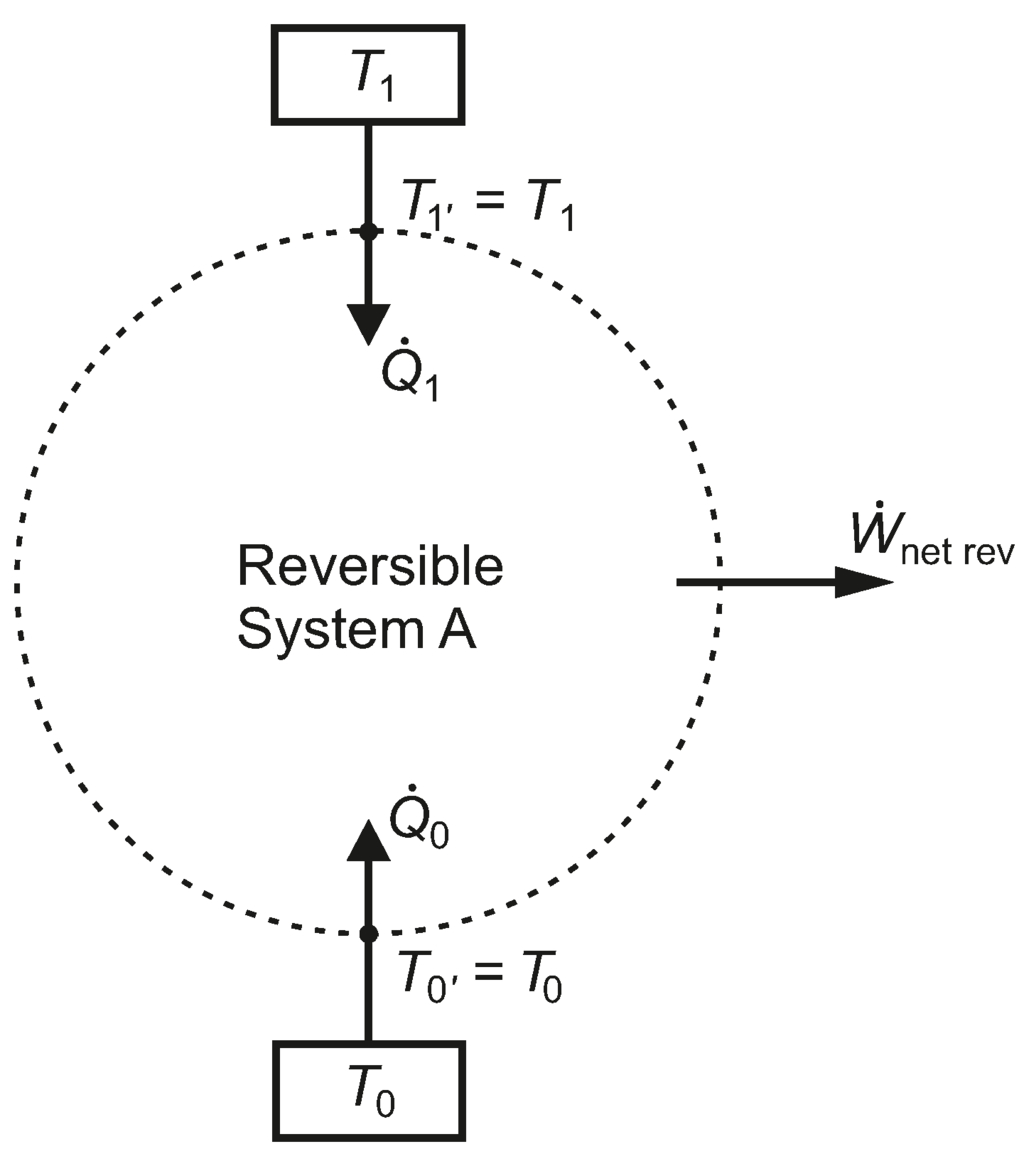
“First law of thermodynamics: The net change in total energy of a system (∆E) is equal to the heat added to the system (Q) minus work done by the system (W).” In terms of heat, work and internal energy, the statement/definition of first law of thermodynamics can be stated as Now for the mathematical form, the equation of first law of thermodynamics includes heat (Q), work (W), and internal energy (U). “Energy can neither be created nor can be destroyed but transformation of one form into another form can be possible.” In previous section, we saw that 1st law of thermodynamics is nothing but a law of conservation of energy. Here I will explain you the entire concept hidden in the equation of 1st law of thermodynamics. Infact the example which I have mentioned in this explanation makes me remember the first law very easily. Now I have found the best way to remember this equation of first law of thermodynamics. When I was a student, I faced many problems in understanding the mathematical form of first law of thermodynamics.īut then I read from books, internet and I also learned from my teacher. Summary (Read this if you are in hurry).Why is absolute zero not possible? (simple).What is absolute zero temperature? Is it possible to achieve?.Entropy statement of Second law of thermodynamics.Second law of thermodynamics based on entropy.Second law of thermodynamics for heat pump/refrigerator (Clausius’s statement).How to remember Kelvin Planck’s statement?.Second law of thermodynamics for heat engine (Kelvin Planck’s statement).Why do we use the second law of thermodynamics? Equation for first law of thermodynamics.So many energies in a single picture (short and sweet).Application of zeroth law of thermodynamics.What did you find from the zeroth law of thermodynamics?.What is the zeroth law of thermodynamics?.Why is there a zeroth law of thermodynamics?.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
